Rui Yuan, Yuan-jiang Wang, Yue Fang, Wen-hui Ge, Wei Lin, Ming-qi Li, Jiang-biao Xu, Yu Wan, Yun Liu, Hui Wu*, The First Direct Synthesis of Chiral Tröger's Bases Catalyzed by Chiral Glucose-containing Pyridinium Ionic Liquids, Chem. Eng. J. 316, 1026, 2017.
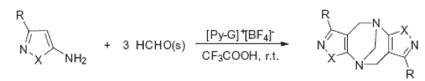
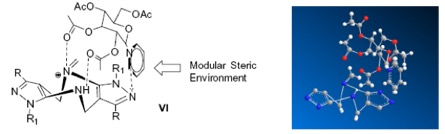
Ab s t r a c t:Tröger’s base (TB) and its derivatives, especially chiral TBs are widely used in many fields. Although there is an urgent demand for the direct synthesis of pure enantiomers of TBs, convenient synthetic strategies are far less developed. Ionic liquids are green and recyclable solvent, therefore, in this paper, novel chiral glucose-containing pyridinium ionic liquids (1-((2S,3S,4R,5S,6S)-3,4,5-triacetoxy-6-(acetoxymethyl)tetra hydro-2H-pyran-2-yl)pyridin-1-ium tetrafluoroborate, [Py-G]+[BF4]-) was synthesized and used as catalyst and solvent, while trifluoroacetic acid (TFA) was used as co-catalyst, to promote the one-step direct asymmetric synthesis of Tröger’s bases with pyrazol flank firstly at room temperature with high yields (up to 83%) and high ee value (up to 84%). A reasonable reaction mechanism was investigated by using chemical experiments, 1H NMR analysis and mass spectrometry. The novel method is environmental friendly and the result indicated its value of practical application.
Highlights:
1.Novel chiral glucose-containing pyridinium ionic liquids were synthesized.
2.The new chiral ionic liquids were used as catalyst and solvent.
3.The direct asymmetric synthesis of Tröger’s base derivatives was put forward firstly.
4.The reasonable reaction mechanism was carefully investigated.
5.The strategy is green, cost-effective, operationally convenient and highly efficient.
Chem. Eng. J. is one of the top journals in engineer and technology.

